|
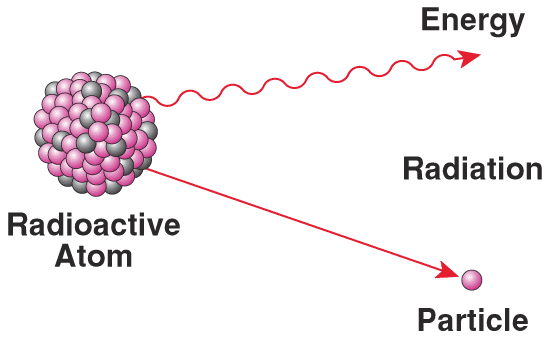
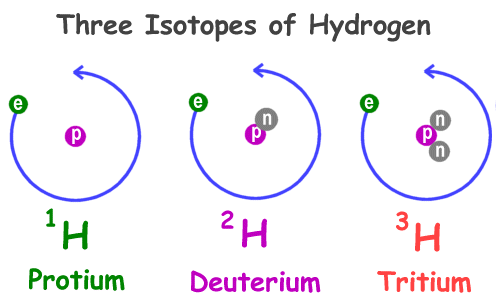
All atoms found in nature are either stable or unstable. Isotopes of a single element vary in the number of neutrons in their nuclei. Since they still have the same number of protons, all these isotopes of an element have identical chemical behavior. However, since they have different numbers of neutrons, the isotopes of the same element may have different radioactivity. These isotopes can be called either radioisotopes or radionuclides.
Isotopes have the same number of protons in their nucleus (as to make them the same element), but a varying number of neutrons. They become radioisotopes when the atoms contain an unstable combination of protons and neutrons. The unstable atom gets rid of excess energy by emitting radiation, in the forms of alpha, beta and gamma rays. Many radionuclides occur naturally, and others are produced artificially. |